The world of complex chemical compounds is vast and intricate. These compounds play crucial roles in various industries, including pharmaceuticals, agriculture, and materials science. According to the Chemical Abstracts Service, complex chemicals represent a significant portion of all chemical research activities. Expert Dr. Emily Chen notes, “Understanding complex chemicals is fundamental for innovation in sustainable solutions.”
Various complex chemical compounds have diverse applications. For instance, they are used in creating advanced materials with unique properties, often unparalleled in performance. However, the complexity of these compounds also brings challenges in synthesis and application. Researchers are constantly seeking improvements to make these processes more efficient. The journey of innovation in the complex chemical field is not always smooth. It often highlights the need for better safety measures and environmental considerations.
As we explore the ten best complex chemical compounds, we will encounter both their remarkable benefits and the obstacles researchers face. The balance between safety and advancement is delicate. This balance is crucial in moving forward in the ever-evolving science of complex chemicals.

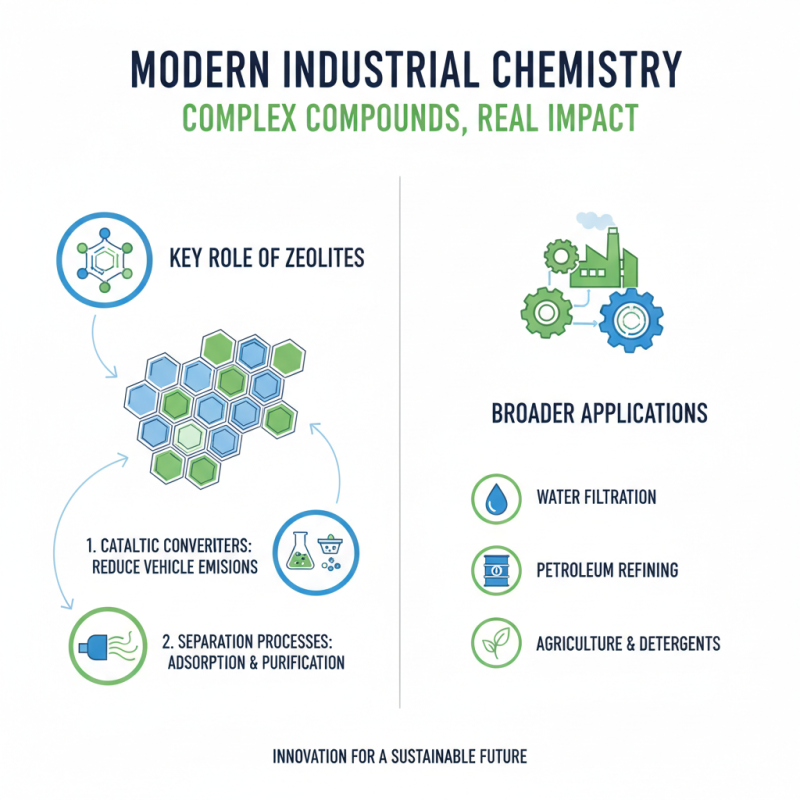
In modern industry, complex chemical compounds play crucial roles. These compounds are not just scientific curiosities; they have real-world applications that impact daily life and various sectors. One significant example is zeolites. These crystalline materials are used in catalytic converters. They help reduce harmful emissions from vehicles. Their ability to adsorb molecules makes them invaluable in various separation processes.
Another notable compound is metal-organic frameworks (MOFs). These materials are known for their high surface area. They are used in gas storage and separation. Their design can be tailored for specific applications. Yet, the scaling up of MOFs for commercial use remains a challenge. Their production can be complex and costly, raising questions about sustainability.
Chelating agents are also important. They bind metal ions, preventing them from precipitating. This property is vital in water treatment. However, their environmental impact continues to be a topic for discussion. Each of these compounds shows promise, but their development often encounters hurdles. Balancing efficiency, cost, and environmental concerns is not straightforward.
Complex chemical compounds play a significant role in the field of pharmaceuticals. They consist of multiple elements and often exhibit unique properties. This complexity allows for diverse applications in drug formulation.
For instance, metal-organic frameworks can transport drugs more efficiently. Their porous structure enhances the delivery of therapeutic agents.
Some complexes have specific targets in the body. These compounds can bind selectively to receptors, providing targeted therapy. They can improve drug solubility and stability. This is crucial for many medications that require precise dosing. However, the preparation of these compounds can be complicated. Consistency and reproducibility remain challenges in their production.
Additionally, complex compounds are often used in diagnostics. They can act as contrast agents in imaging techniques. This application helps in identifying diseases early. However, safety concerns arise. Some compounds may cause side effects. Continuous research is necessary to improve their efficacy and safety profiles. The journey of developing these compounds is filled with trials and errors.
Coordination complexes play a crucial role in environmental chemistry. They help in the removal of harmful pollutants from soil and water. These compounds can bind to metal ions and toxins, which reduces their toxicity and mobility. For example, chelating agents are used to extract heavy metals from contaminated sites.
There are unique properties of these complexes that enhance their effectiveness. Metal ions can stabilize various environmental conditions. This allows for better degradation of pollutants. However, the treatment processes can be costly and sometimes inefficient. Not all complexes work the same way in diverse environments.
Tips: Regularly assess local contaminants to identify the most suitable remediation strategy. Combining different complexing agents might lead to better results. Always consider the potential environmental impact of your chosen methods.
Complex compounds play a pivotal role in catalysis, driving innovations in various chemical processes. Their unique structures allow for specific interactions with reactants. This leads to enhanced reaction rates and improved selectivity. Research shows that certain metal complexes outperform traditional catalysts in efficiency. These advancements are crucial for developing greener and more sustainable chemical practices.
While the efficiency of complex compounds is evident, challenges remain. For instance, the stability of some catalysts can be a concern. Environmental conditions may affect their performance. Additionally, the complexity in synthesis can hinder practical applications. A careful evaluation of these aspects is necessary.
Despite these setbacks, the future looks promising. Innovations in ruthenium and palladium complexes are emerging. The focus on refining the synthesis processes is a positive step forward. These developments suggest that the potential of complex compounds in catalysis is far from fully realized. There’s much to learn from both successes and failures in this dynamic field.

Organometallic compounds play a crucial role in industrial chemistry, serving as building blocks for various synthesis processes. These compounds often contain carbon-metal bonds, enabling unique reactivity. Numerous industries utilize organometallics for catalyst development, polymer production, and pharmaceuticals. For example, the global organometallic compounds market is projected to reach over $25 billion by 2025, driven by increasing demand in catalysis and materials science.
In synthesis, organometallics allow for selective reactions. They enable the formation of carbon-carbon and carbon-heteroatom bonds, making them essential for constructing complex molecules. The versatility of organometallic catalysts can dramatically increase efficiency in chemical processes. This efficiency often leads to lower energy consumption and reduced waste. However, challenges remain in terms of scalability and cost, which require ongoing research and development.
Tips: Always consider safety when handling organometallic compounds. They can be sensitive to moisture and air. Additionally, some reactions require specific conditions to achieve optimal results. Continual adjustments may be necessary to refine processes. Keeping records of each trial can lead to significant improvements and insights over time. Staying informed about new advancements in the field can also open doors to innovative applications.





Contact our team with questions, product inquiries or challenge us to engineer a solution for you.
Tel: +1 716 433 6764
Fax: +1 716 433 2850
Email: sale@ashymed.com
VanDeMark Chemical Inc.
One North Transit Road
Lockport, NY 14094 USA